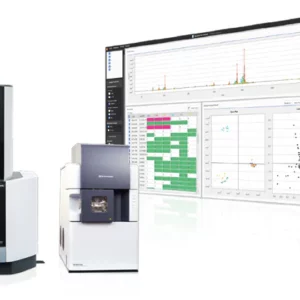
QC Reporter
Compatible with the MALDI-8020 benchtop linear MALDI-TOF mass spectrometer, this software provides a simple and secure platform for typical QC workflows.
- Описание
- Features
Описание
QC Reporter™: mass confirmation and quality assessment with confidence
Within a given manufacturing process, quality control (QC) plays a fundamental role in guaranteeing the supply of a high quality product. QC Reporter is an automated analysis and reporting software solution for QC environments. Compatible with the MALDI-8020 benchtop linear MALDI-TOF mass spectrometer, this software provides a simple and secure platform for typical QC workflows.
QC Reporter
QC Reporter is a fully automated software application designed for high-throughput quality control analysis using MALDI-TOF MS. The acquisition, calibration, processing, quality assessment and reporting processes are performed automatically within a single platform and a simple, intuitive workflow design. Using QC Reporter, a typical QC experiment can be performed with a few simple steps:
• Defining the acquisition and quality assessment criteria settings
• Importing samples for analysis using a predefined sample worklist
• Viewing and/or exporting the results
The quality criteria (detection of the target mass and amount of permitted adducts/impurities) are applied on the fly for each acquired sample and are presented in a colour-coded manner providing an at a glance summary for easy reading. The option to subsequently export a summary of the entire experiment or to generate individual sample reports provides the user with options for different workflows.
Key features:
— Security via individual user login and Administrator control of user roles and permissions
— Flexibility:
applicable to different applications (peptides, proteins, oligonucleotides and small molecules)
— Fully compatible with the MALDI-8020 mass spectrometer
— Simple operation:
set the acquisition and criteria parameter sets, run the samples and view the results
— Intuitive user-interface:
colour-coded results providing an at a glance summary of the QC experiment. Review individual results via the interactive slide graphic
— Comprehensive reporting:
all relevant information is presented in exportable sample reports
— Seamless relationship with MALDI Solutions Data Acquisition software via the MALDI Solutions database
Features
QC criteria
A wide selection of user-defined parameters can be configured as acceptance criteria for the QC assay, providing flexibility for the user to customize each type of experiment for a given application.
Acquisition
The information regarding each sample is provided via a simple .csv spreadsheet containing
Reporting
During an experiment, the acquired spectrum and result for each analyzed sample are displayed in realtime on the target slide graphic using the colour-coded results reporting schema. At the end of the experiment, the results are presented as a tabulated, colour-coded summary report in the main results window.
Data security
User-access to QC Reporter is via password-protected login and is controlled by a System Administrator who manages access within the application. The Administrator is able to control access to features within the software via various roles which can be applied to individual user accounts.
Features
QC criteria
A wide selection of user-defined parameters can be configured as acceptance criteria for the QC assay, providing flexibility for the user to customize each type of experiment for a given application. These include:
• Mass tolerance (compared with the expected mass)
• Signal-to-noise threshold
• Minimum resolution of the target mass
• Acceptable limits for adducts
• Acceptable limits for impurity levels
Acquisition
The information regarding each sample is provided via a simple .csv spreadsheet containing:
• Sample location on the MALDI target
• Sample identifier/sample name
• Expected molecular weight
• Acquisition/calibration sample analysis methods (parameter sets)
• Additional comments (optional)
The acquisition, calibration and data processing methods are created and saved in the MALDI Solutions™ Data Acquisition application and can be retrieved via the seamless integration with the shared SQL MALDI Solutions database. Experiments and results are stored in the SQL database and benefit from the database security features designed to limit access to data/results and prevent unauthorized deletion of data. The data for each sample is evaluated and compared against the QC criteria, specified in the acquisition parameter set, in realtime. As the experiment progresses, the target slide graphic is updated with a colour-coded indication of the obtained results:
Green = Pass: the expected mass has been found within the given tolerance and all other specified criteria are within the acceptable limits
Red = Fail: the expected mass has not been found within the given tolerance or other specified criteria were set to trigger a ‘Fail’ result
Yellow = Query: the expected mass has been found but it does not meet the absolute criteria (e.g. signal-to-noise, resolution) or high level of impurities/adducts have been detected
Reporting
During an experiment, the acquired spectrum and result for each analyzed sample are displayed in realtime on the target slide graphic using the colour-coded results reporting schema. At the end of the experiment, the results are presented as a tabulated, colour-coded summary report in the main results window. From this view, the user can further interrogate individual sample results.
After reviewing the results, these can be exported as PDF printable reports for the experiment summary, all samples or selected individual samples. These can be used to report the results back to customers or kept as records of the sample analysis jobs.
Data security
User-access to QC Reporter is via password-protected login and is controlled by a System Administrator who manages access within the application. The Administrator is able to control access to features within the software via various roles which can be applied to individual user accounts.
For example, the acquisition, calibration and data processing methods are created and/or edited by the Administrator or by users who have been granted this permission. This protects the acquisition methods against unpermitted changes and provides peace of mind in regulated environments.